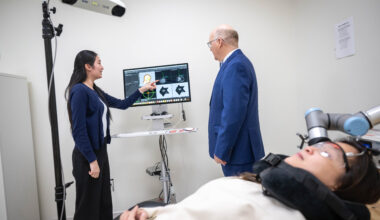
The ALS Society of British Columbia (ALSBC), in partnership with the UBC ALS & Related Disorders (ALSRD) Research Program, is proud to announce the formal return of ALS clinical trials to British Columbia after nearly 15 years.
Under the leadership of Dr. Erik Pioro, research staff training and site initiation preparations are now complete. The Phase 2 trial ALSTARS study, which is testing the COYA 302 drug, is now actively recruiting candidates at the UBC ALSRD Clinic, located at the Djavad Mowafaghian Centre for Brain Health. This randomized, double-blind, placebo-controlled, multi-centre trial will run for 24 weeks, followed by an additional 24-week blinded active extension to evaluate the drug’s safety and efficacy.
“It’s a momentous day for the ALS community in British Columbia,” says Donald Miyazaki, Executive Director of the ALS Society of BC. “ALSBC Project Hope started with a mission of bringing clinical trials to our research program’s new home at UBC so patients can once again contribute to research in their own backyards. Through our partnership with the Province of British Columbia, UBC, and thousands and patients, caregivers, families, and donors who have made this dream possible, we thank you for helping us achieve this milestone. This is another step towards our vision of living in a world without ALS.”
The ALSTARS trial will be conducted at approximately 20-25 sites in Canada and the United States and will evaluate the safety and efficacy of an investigational treatment for adults with ALS.
COYA 302 is an experimental therapy that combines two medicines to help calm harmful inflammation in the body. It works by boosting the activity of certain immune cells that reduce inflammation, while also limiting the activity of other immune cells that can make inflammation worse.
Participants will be randomly assigned to receive one of two regimens of COYA 302 or placebo for 24-weeks in the double-blind period. Those who complete this phase may be eligible to continue in a 24-week blinded active extension.
The study will assess disease progression using established ALS clinical outcome measures, including the ALS Functional Rating Scale-Revised (ALSFRS-R), neurofilament levels (NfL), maximal inspiratory pressure (MIP), slow vital capacity (SVC) and neurological assessments. Additional objectives include evaluating biomarkers and monitoring safety through routine clinical assessments and adverse event tracking.
“These trials represent far more than individual studies,” says Dr. Pioro. “hey offer renewed hope to patients and families who have long had to travel outside the province for experimental therapies.”
Individuals interested in participating can contact the study coordinator at ALSRDClinicRes@vch.ca or 604-827-2311.
Background information about the study is available on the Coya Therapeutics website, as well as on the ClinicalTrials.gov website, which includes details on the study design and eligibility criteria.
A version of this announcement was originally published on the ALS Society of BC website.